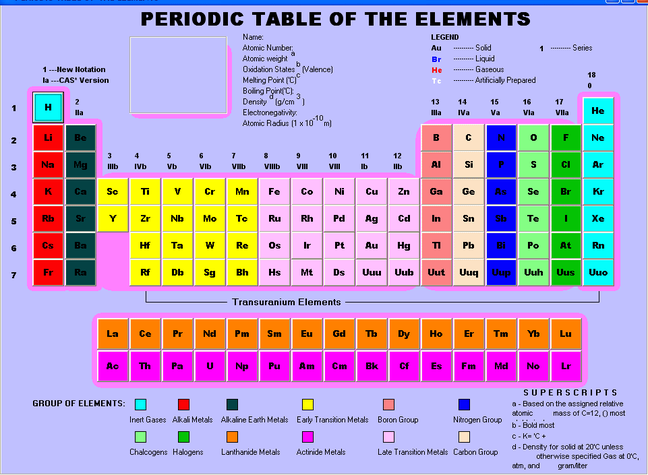
*THE PICTURE ABOVE SHOWS WHERE GROUPS AND PERIODS ARE NUMBERED ON THE PERIODIC TABLE.*
The Modern Periodic Table
The periodic table that we have now has been revised multiple times before it was actually correct. The periodic table is organized in a particular way and it took a few smart scientists to continuously edit and make changes to it, in order for it to be the way it is now. The periodic table is arranged in groups, periods, by valence electrons, and orbitals.
Groups:
A group is a column of elements. There are 18 different groups in the periodic table, and the elements in each group are arranged in the order in which they have similar physical or chemical characteristics in their outermost electron shells within their atoms. There are 3 systems of group numbering.
Periods:
A period is a row of elements. there are 7 periods in periodic table and the elements in each period all have the same number of electron shells. With each group across a period, the elements have 1 more proton (and electron) and become less metallic.
Valence Electrons:
A valence electron is the electron in the outermost electron shell of an atom. Valence electrons can participate in the formation of chemical bonds, and the presence of valence electrons can determine the chemical properties of an element and whether or not it's able to bond with other chemicals. An atom with a closed shell of valence electrons is often chemically inert. Valence electrons have the ability to absorb/release energy. The number of valence electrons can be found out by the group that the element is in on the periodic table. The number in the unit's place can be associated with a neutral atom of an element listed under that particular column.
The Modern Periodic Table
The periodic table that we have now has been revised multiple times before it was actually correct. The periodic table is organized in a particular way and it took a few smart scientists to continuously edit and make changes to it, in order for it to be the way it is now. The periodic table is arranged in groups, periods, by valence electrons, and orbitals.
Groups:
A group is a column of elements. There are 18 different groups in the periodic table, and the elements in each group are arranged in the order in which they have similar physical or chemical characteristics in their outermost electron shells within their atoms. There are 3 systems of group numbering.
Periods:
A period is a row of elements. there are 7 periods in periodic table and the elements in each period all have the same number of electron shells. With each group across a period, the elements have 1 more proton (and electron) and become less metallic.
Valence Electrons:
A valence electron is the electron in the outermost electron shell of an atom. Valence electrons can participate in the formation of chemical bonds, and the presence of valence electrons can determine the chemical properties of an element and whether or not it's able to bond with other chemicals. An atom with a closed shell of valence electrons is often chemically inert. Valence electrons have the ability to absorb/release energy. The number of valence electrons can be found out by the group that the element is in on the periodic table. The number in the unit's place can be associated with a neutral atom of an element listed under that particular column.
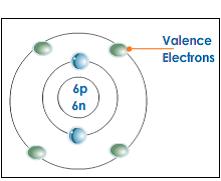
*this picture shows what valence electrons look like in an atom*
Orbitals:
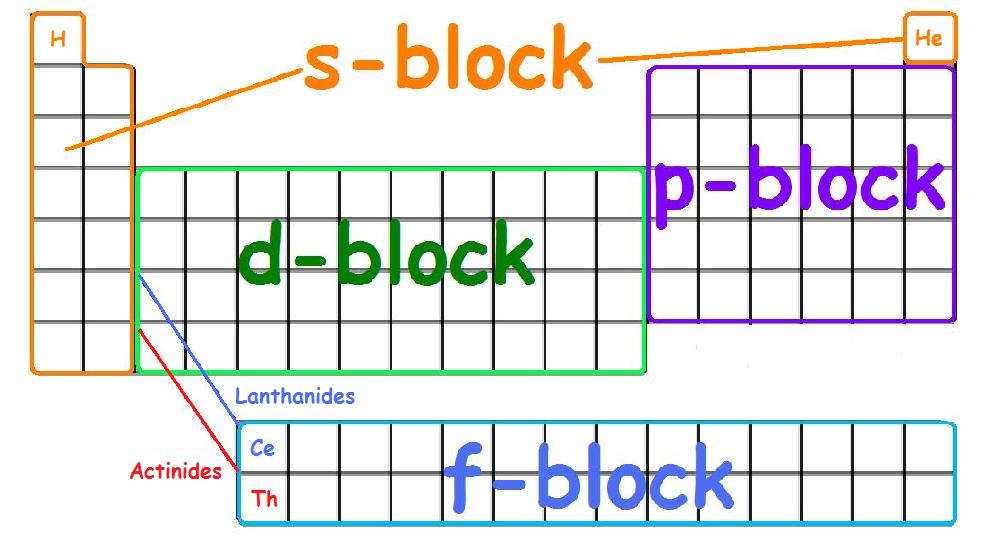
An orbital is the region of space that an electron habits. There are four kinds of orbitals:
S, P, D, and F. The picture below shows where each orbital occupies which groups on the periodic table.
An orbital is the region of space that an electron habits. There are four kinds of orbitals:
S, P, D, and F. The picture below shows where each orbital occupies which groups on the periodic table.